|
Hydrogen is the most abundant chemical substance in the universe, constituting roughly 75. It is colorless, odorless, tasteless, 9 non-toxic, and highly combustible. It is the lightest element and, at standard conditions, is a gas of diatomic molecules with the formula H2. Since the mass of a neutron and the mass of a proton are both very close to 1 u, the atomic mass of an isotope is often nearly the same as its mass number. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals. Hydrogen is a chemical element it has symbol H and atomic number 1. An isotope's mass number is closely related to its atomic mass, which is the mass of the isotope expressed in units of u. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The unity for atomic mass is gram per mol. The lightest chemical element is Hydrogen and the heaviest is Hassium. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight).  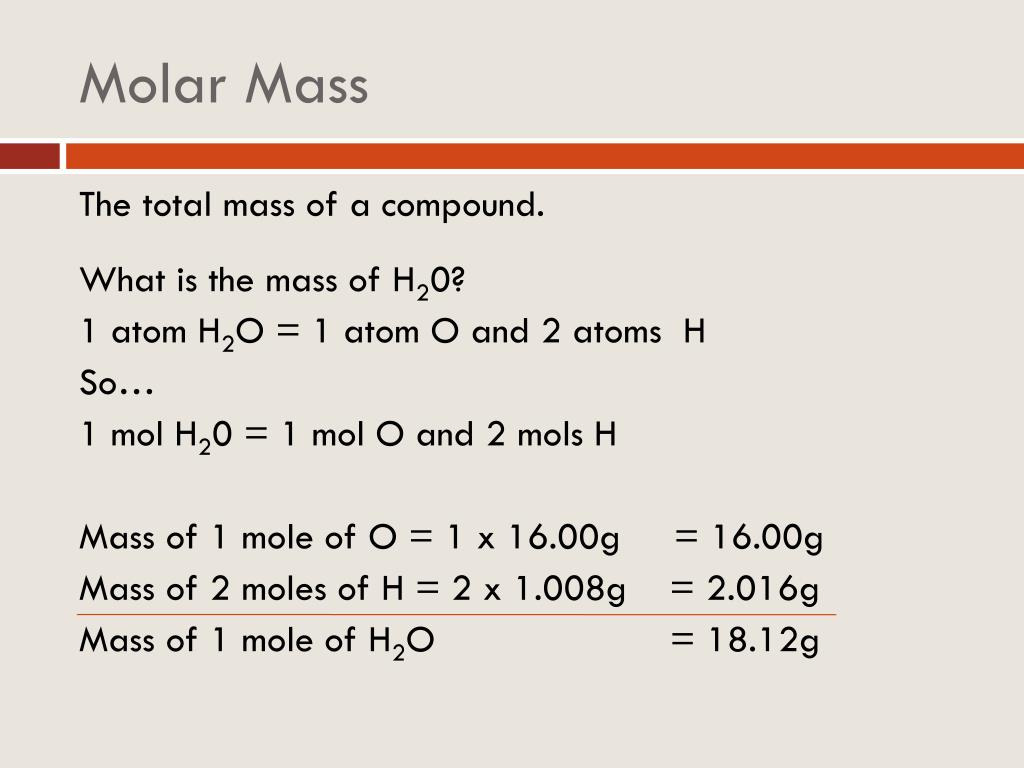
This list contains the 118 elements of chemistry. If the mass of a substance is known, the number of moles in the substance can be calculated. Separation and Concentration Purification RequestĬhemical elements listed by atomic mass The elements of the periodic table sorted by atomic massĬlick on any element's name for further information on chemical properties, environmental data or health effects. The molar mass of an element is found on the periodic table, and it is the element's atomic weight in grams/mole (g/mol).Plant Inspection & Process Optimalisation.The former definition of the mole was that a mole was 12 g of 12C contains 1 mole of 12C atoms (its molar mass is 12 g/mol). Both can be used to solve problems that would be hard to do by eye. Practice what you’ve learned and study for the AP Chemistry exam with more than 100 AP-aligned questions. Learn about moles and molar mass, mass spectrometry, electron configurations, periodic trends, and more. 
It is a common element in the universe, estimated at seventh in total abundance in the Milky Way and the Solar System. 
Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. The first conversion factor can be used to convert from mass to moles, and the second converts from moles to mass. This unit explores the atomic theory of matter, the foundational premise of chemistry. Nitrogen is a chemical element it has symbol N and atomic number 7. Per the amu definition, a single 12C atom weighs 12 amu (its atomic mass is 12 amu). 1 molAl 26.98g Al and 26.98gAl 1mol Al (5.4.1) (5.4.1) 1 m o l A l 26.98 g A l a n d 26.98 g A l 1 m o l A l. Options for hiding the symbol or name of the elements provide a handy learning aid for memorizing the periodic table. (credit: modification of work by Mark Ott).īecause the definitions of both the mole and the atomic mass unit are based on the same reference substance, 12C, the molar mass of any substance is numerically equivalent to its atomic or formula weight in amu. Use this periodic table for calculating molar mass for any chemical formula. The molar mass has units of g/mol or kg/mol. A mole of substance is equal to Avogadros number (6.023×10 23) of that substance. From left to right (bottom row): 32.1 g sulfur, 28.1 g silicon, 207 g lead, and 118.7 g tin. The molar mass is the mass of one mole of substance, whether the substance is an element or a compound. From left to right (top row): 65.4 g zinc, 12.0 g carbon, 24.3 g magnesium, and 63.5 g copper. In this movie, we show how to calculate the molecular weight of a substance from the atomic weights given on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
